COVID-19 Over-The-Counter (OTC) Antigen Testing
Antigen tests are non-prescribed rapid tests that detect genetic material, called RNA, from the virus that causes COVID-19 (SARS-CoV-2). A specimen sample is collected from the nose or mouth and results are produced within 10-15 minutes. To learn more, visit CDC interim guidance.
COVID-19 Over-the-Counter (OTC) antigen tests are an easy and convenient way to test for COVID-19. These tests can be taken anywhere and are often called “home tests” or “at-home tests”. The California Department of Public Health (CDPH) has validated the use of OTC antigen test kits even beyond the printed or FDA-authorized listed expiration dates. For more information, please refer to the tables on the right.
Please use the page to learn more about antigen testing and to find out where you can acquire OTC Antigen Tests for at-home use on this page.
Please note, these should be considered options and not a comprehensive list of all locations and businesses offering COVID-19 OTC antigen tests.
For questions or assistance, call our COVID-19 Hotline at
(909) 387-3911, Monday – Friday from 9 a.m. – 5 p.m.
| FDA Expiration Date Extension Information |
|---|
| If you would like to see a list of the latest FDA Expiration Date Extensions, please see below: • iHealth® Per the California Department of Public Health (CDPH), replace your tests with fresh ones as the extended expiration dates approach. If you have to, you can use an expired test until you can get new ones, as long as the control line is working. See your test instructions for details about the control line. |
Find Over-the-Counter (OTC) Antigen Test
Free at-home antigen self-test kits are available for pickup at County COVID-19 sites.
Limit two kits per person. Exceptions are available for caretakers or individuals who are homebound.
Ontario – Transitional Assistance Department (TAD)
1627 E. Holt Blvd., Ontario
Monday – Friday from 9 a.m. – 4 p.m.
Rancho Cucamonga – Transitional Assistance Department (TAD)
10825 Arrow Rte., Rancho Cucamonga
Monday – Friday from 9 a.m. – 4:30 p.m.
Redlands – Transitional Assistance Department (TAD)
1811 W. Lugonia Ave., Redlands
Monday – Friday from 9 a.m. – 4 p.m.
**Location: TAD Main Entrance Lobby**
Rialto – Transitional Assistance Department (TAD)
1175 W. Foothill Blvd., Rialto
Monday – Friday from 9 a.m. to 4 p.m.
Victorville WIC
15247 Eleventh St., Ste.700, Victorville
Monday – Thursday from 9 a.m. – 4 p.m.
Friday from 8 a.m. – 3 p.m.
**Location: Lobby**
As of February 22, 2022, OTC Antigen test kits are available at every County Library free of charge. Please visit: sbclib.org for additional information.
- Apple Valley Library
14901 Dale Evans Parkway, Apple Valley 92307
Phone: (760)247-2022
Type of Test(s) Offered: Over-the-Counter (OTC) Antigen take home - Cal Aero Preserve Academy Branch
15850 Main St., Chino 91708
Phone: (909) 606-2173
sbclib.org/cal-aero-preserve
Type of Test(s) Offered: Over-the Counter (OTC) Antigen Test - Chino Branch Library
13180 Central Ave., Chino 91710
Phone: (909) 465-5280
sbclib.org/chino
Type of Test(s) Offered: Over-the Counter (OTC) Antigen take home - Chino Hills James S. Thalman Branch Library
14020 City Center Dr., Chino Hills 91709
Phone: (909) 590-5380
sbclib.org/james-s-thalman
Type of Test(s) Offered: Over-the counter (OTC) Antigen take home - Crestline Branch Library
24105 Lake Gregory Dr., Crestline 92325
Phone: (909) 338-3294
sbclib.org/crestline
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home - Fontana Lewis Library & Technology Center
8437 Sierra Ave., Fontana 92335
Phone: (909) 574-4500
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home - Kaiser Branch
11155 Almond Ave., Fontana 92337
Phone: (909) 357-5900
sbclib.org/kaiser
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home - Summit Branch
15551 Summit Ave., Fontana 92336
Phone: (909) 357-5950
sbclib.org/summit
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home - Grand Terrace Branch Library
22795 Barton Rd., Grand Terrace 92313
sbclib.org/grand-terrace
Phone: (909) 783-0147
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home - Hesperia Branch Library
6950 7th Ave., Hesperia 92345
Phone: (760) 244-4898
sbclib.org/hesperia
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home - Highland Sam J. Racadio Library & Environmental Learning Center
7863 Central Ave., Highland 92346
Phone: (909) 798-8490
sbclib.org/highland
Type of Test(s) Offered: Over-the-Counter (OTC) Antigen take home - Joshua Tree Branch Library
6465 Park Blvd., Joshua Tree 92407
Phone: (760) 336-8615
sbclib.org/joshua-tree
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home - Lake Arrowhead Branch
27235 Highway 189, Bluejay 92317
Phone: (909) 337-3118
sbclib.org/lake-arrowhead
Type of Test(s) Offered: Over-the-counter (OTC) Antigen Test - Loma Linda Branch Library
25581 Barton Rd., Loma Linda 92345
Phone: (909) 796-8681
sbclib.org/loma-linda
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- Lucerne Valley Janice Horst Branch
33103 Old Woman Springs Rd., Lucerne Valley 92356
Phone: (760) 248-7521
sbclib.org/lucerne-valley
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- Mentone Senior Center and Library
1131 Opal Ave., Mentone 92359
Phone: (909) 794-0327
sbclib.org/mentone
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- Muscoy Baker Family Learning Center
2818 Macy St., Muscoy 92407
Phone: (909) 887-5167
sbclib.org/baker-family
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- San Bernardino County Library- Montclair
9955 Fremont Ave., Montclair 91763
Phone: (909) 387-3911
sbclib.org/montclair
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- Needles Branch Library
1111 Bailey Ave., Needles 92363
Phone: (760) 326-9255
sbclib.org/needles
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- Phelan Memory Library
9800 Clovis Rd., Phelan 92371
Phone: (760) 868-3053
sbclib.org/phelan-memorial
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- Carter Branch Library
2630 N. Linden Ave., Rialto 92376
Phone: (909) 854-4100
sbclib.org/carter
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- Rialto Branch Library
251 W. 1st St., Rialto 92376
Phone: (909) 875-0144
sbclib.org/rialto
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- Running Springs Library
2677 Whispering Pines Dr., Running Springs 92382
Phone: (909) 867-3604
sbclib.org/running-springs
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- Trona Library
82805 Mountain ave., Trona 93562
Phone: (760) 372-5847
sbclib.org/trona
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- Twentynine Palms Branch Library
6078 Adobe RD., Twentynie Palms 92277
Phone: (760) 367-9519
sbclib.org/twentynine-palms
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- Wrightwood Branch Library
6011 Pine St., Wrightwood 92397
Phone: (760) 249-4577
sbclib.org/wrightwood
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- Yucaipa Branch
12040 5th St., Yucaipa 92399
Phone: (909)790-3146
sbclib.org/yucaipa
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
- Yucca Valley Branch Library
57271 29 Palms Hwy., Yucca Valley 92284
Phone: (760) 228-3244
sbclib.org/yucca-valley
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
To find locations near you to purchase COVID-19 OTC antigen tests, please select one of the following options:
- Queens Pharmacy
Address: 18522 Highway 18, Ste. 103 Apple Valley, 92307
Phone: (760) 946-4700
Website: mygnp.com/pharmacies/queens
Type of Test(s) Offered: Over-the-Counter (OTC) Antigen take home
Cost: Free with insurance or $12.99
- Sav-on Pharmacy
Address: 20261 Hwy 18, Apple Valley 92307
Phone: (760) 242-1323
Website: www.albertsons.com/pharmacy.html
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with insurance or $12
- Teplow Drugs
Address: 404 E. Main St., Barstow 92311
Phone: (760) 256-2726
Type of Test(s) Offered: Over-the-Counter (OTC) Antigen take home
Cost: $8.78
- Croal’s Rexall Drugs
Address: 901 E. Williams St., Barstow 92311
Phone: (760) 256-8968
Type of Test(s) Offered: Over-the-Counter (OTC) Antigen take home
Cost: $15
- Procare Pharmacy
Address: 18671 Valley Blvd., Bloomington 92316
Phone: (909) 990-3000
Type of Test(s) Offered: Over-the-Counter (OTC) Antigen take home
Cost: Free with Insurance or $12-$20
- VIP Pharmacy
Address: 13193 Central Ave., #120, Chino 91710
Phone: (909) 573-0123
Website: iepharmacy.com
Type of Test(s) Offered: Over-the-Counter (OTC) Antigen take home
Cost: Free with Insurance or $12.99
- Santa Fe Pharmacy
Address: 12916 Central Ave., Chino 91710
Phone: (909) 342-9181
Website: santafepharmacy.com
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $19.99
- Chino Medical Pharmacy
Address: 5365 Walnut Ave., # D, Chino 91710
Phone: (909) 591-6038
Website: chinomedicalpharmacy.com
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: $14.99-27.99
- Chino Care Pharmacy
Address: 13085 Central Ave., #2, Chino 91710
Phone: (909) 927-8323
Website: chinocarerx.com
Type of Test(s) Offered: Over-the-Counter (OTC) Antigen take home
Cost: Free with Insurance or $20
- K’s Pharmacy
Address: 12582 Central Ave., Chino 91710
Phone: (909) 591-7429
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $12-$15
- CaliCare Pharmacy
Address: 12144 Central Ave., Chino 91710
Phone: (909) 591-8444
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $40
- Chino Valley Community Health Center (Look-Alike, Primary)
Address: 13193 Central Ave., Chino 91710
Phone: (909) 464-9675
Monday-Friday 9a.m.-5p.m
*Please call the health center to confirm testing availability and procedures prior to arrival.
Type of Test(s) Offered: Rapid Antigen
Cost: Free with Insurance
- TotalCare
Address: 13768 Roswell Ave., Chino 91710
Phone: (909) 591-8200
Type of test(s) Offered: POC Rapid Antigen
Cost: Free with insurance or $80-$165
- Chino Hills Professional Pharmacy
Address: 2140 Grand Ave., #130, Chino Hills 91709
Phone: (909) 364-9248
Website: www.chinohillsprofessionalpharmacy.com
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with insurance or $14.50
- Sav-on Pharmacy
Address: 3255 Grand Ave., Chino Hills 91709
Phone: (909) 590-8373
Website: www.albertsons.com/pharmacy.html
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $12
- Sav-on Pharmacy
Address: 15970 Los Serranos City Club Dr., Chino Hills 91709
Phone: (909) 606-8185
Website: www.albertsons.com/pharmacy.html
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $12
- Cooley Health Pharmacy
Address: 1280 E. Cooley Dr., Ste. 23, Colton 92324
Phone: (909) 514-1730
Website: mygnp.com/pharmacies
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $9.99-$29.99
- Larson’s Pharmacy
Address: 142 W. H St., Colton 92324
Phone: (909) 825-1950
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: $23.49
- Colton Urgent Care
Address: 1181 N. Mount Vernon Ave., Colton 92324
Phone: (909) 639-8800
Website: Coltonurgentcare.com
Type of Test(s) Offered: Rapid Antigen and PCR Lab
Cost: Free with Insurance or $100-$150
- Brotherhood Pharmacy
Address: 1040 S. Mount Vernon Ave., Ste. E, Colton 92324
Phone: (909) 222-4884
Website: brotherhoodpharmacy.refillquick.com
Type of Test(s) Offered: Over-the-Counter (OTC) Antigen take home
Cost: Free with Insurance or $9
- Lake Gregory Pharmacy
Address: 580 Forest Shade, Rd. #7, Crestline 92325
Phone: (909) 338-1875
Website: lakegregorypharmacy.com
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: $15
- Primera Care Pharmacy
Address: 17500 Foothill Blvd., Ste. 7a., Fontana 92335
Phone: (909) 222-6944
Website: mygnp.com
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with insurance $15-$25
- St. Helen Pharmacy
Address: 17577 Arrow Blvd., #101, Fontana 92335
Phone: (909) 428-9842
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $25.50
- St. Mina Pharmacy
Address: 16701 Valley Blvd., Fontana 92335
Phone: (909) 823-2515
Website: www.stminapharmacy.com
Type of Test(s) Offered: PCR Lab and Over-the-Counter (OTC) Antigen take home
Cost: Free with Insurance or $15-$25
- Vons Pharmacy
Address: 7390 Cherry Ave., Fontana 92336
Phone: (909) 429-3933
Website: www.vons.com/pharmacy.html
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $12
- West Point Medical Center
Address: 7774 Cherry Ave., Ste. A, Fontana 92336
Phone: (909) 355-1296
Website: westpointmedicalcenter.com
Type of Test(s) Offered: PCR Lab
Cost: Free with Insurance or $85
- Vons Pharmacy
Address: 12199 Hesperia Rd., Hesperia 92345
Phone: (760) 241-8384
Website: www.vons.com/pharmacy.html
Type of Test(s) Offered: Over-the-county (OTC) Antigen take home
Cost: Free with Insurance or $12
- Sav-on Pharmacy
Address: 7201 Boulder Ave., Highland 92346
Phone: (909) 425-1194
Website: www.albertsons.com/pharmacy.html
Type of Test(s) Offered: Over-the-Counter (OTC) Antigen take home
Cost: Free with Insurance or $12
- Loma Linda Pharmacy
Address: 25620 Barton Rd., Loma Linda 92345
Phone: (909) 796-8308
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: $40
- Canary Pharmacy
Address: 4950 San Bernardino St., Montclair 91763
Phone: (909) 445-0805
Type of Test(s) Offered: Over–the-counter (OTC) Antigen take home
Cost: Free with Insurance
- COVID Clinic
Address: 2022 E. Airport Dr., Lot F, Ontario 91761
Phone: (877) 219-8378
Website: www.covidclinic.org/ont
Type of Test(s) Offered: Rapid Antigen, Rapid PCR Lab, Point of Care (POC)
Cost: Free with Insurance or $75-$150
- COVID Clinic – Ontario Mills (Drive-Through)
Address: 1 Mills Cir., Ontario 91764
* Located near Skecher’s Factory Outlet.
Phone: (877) 219-8378
Website: www.covidclinic.org/ont
Hours: Monday – Friday from 9 a.m. – 5:00 p.m.
Type of Test(s) Offered: Rapid Antigen, Rapid PCR Lab, Point of Care (POC), Rapid Antibody
Cost: Free with Insurance or $75-$150
- Unicare Community Health Center, INC.
Address: 437 N. Euclid Ave., Ontario 91762
Phone: (909) 457-3603
Website: unicarechc.org
*Drive Through only. Please call to make an appointment and to confirm procedure prior to arrival.
Type of test(s) Offered: Over-the-counter (OTC) Antigen, PCR Lab, POC Antigen
Cost: Free
- DK Pharmacy
Address: 10431 Lemon Ave., Rancho Cucamonga 91730
Phone: (909) 493-1500
Website: www.dkpharmacyrc.com
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with insurance or $12.95
- Green Valley Pharmacy
Address: 8237 Rochester Avenue Ste. 120 Rancho Cucamonga 91730
Phone: (909)980-0999
Website: www.greenvalleyrx.com
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with insurance or $12.50
- Sierra Pharmacy Compounding & Medical Supplies
Address: 8661 Base Line Rd., Rancho Cucamonga 91730
Phone: (909) 989-9800
Website: trymyrx.com
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with insurance or $19.99
- Homes Pharmacy
Address: 9713 Base Line Rd., Rancho Cucamonga 91730
Phone: (909) 989-8688
Website: www.homespharmacy.com
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: $12.95
- Sav-on Pharmacy
Address: 8850 Foothill Blvd., Rancho Cucamonga 91730
Phone: (909) 484-8251
Website: www.albertsons.com/pharmacy.html
Type of Test(s) Offered: Over-the-counter (OTC) Antigen Take Home
Cost: Free with Insurance or $12
- Redlands Urgent Care Center
Address: 301 W. Redlands Blvd., Redlands 92373
Phone: (909) 335-1900
Website: www.redlandsurgentcare.com
Type of Test(s) Offered: Rapid Antigen, PCR Lab, Point of Care (POC)
Cost: Free with Insurance or $150
- Sav-on Pharmacy
Address: 450 E. Cypress Ave., Redlands 92373
Phone: (909) 793-2218
Website: www.albertsons.com/pharmacy.html
Type of Test(s) Offered: PCR Lab, Over-the-counter (OTC) Antigen Take Home
Cost: Free with Insurance or $12
- Vons Pharmacy
Address: 522 N. Orange St., Redlands 92373
Phone: (909) 748-7788
Website: www.vons.com/pharmacy.html
Type of Test(s) Offered: PCR Lab, Over-the-counter (OTC) Antigen Take Home
Cost: Free with Insurance or $12
- Rialto Pharmacy
Address: 1851 N. Riverside Ave., Rialto 92376
Phone: (909) 961-2565
Type of Test(s) Offered: Over-the-counter (OTC) Antigen Take Home
Cost: Free with Insurance or $20
- North Rialto Drugs
Address: 531 Foothill Blvd., Rialto 92376
Phone: (909) 875-2131
Type of Test(s) Offered: Over-the-counter (OTC) Antigen Take Home
Cost: Free with insurance or $19.99
- ABC Pharmacy
Address: 826 Foothill Blvd., Rialto 92376
Phone: (909) 546-1000
Type of Test(s) Offered: POC Rapid Antigen, Over-the-counter (OTC) Antigen Take Home
Cost: Free with Insurance or $9.99-$85
- University Urgent Care
Address: 941 Kendall Dr., Ste. C, San Bernardino 92407
Phone: (909) 726-3200
Website: universityurgentsb.com/?utm_source=GMBSocialClimb&utm_medium=UniversityUrgentCare
Type of Test(s) Offered: POC Rapid Antigen, Rapid PCR Lab, Antibody
Cost: Free with Insurance or $125
- Keystone Industrial Medicine
Address: 1950 S. Sunwest Ln., Ste. 108, San Bernardino 92407
Phone: (909) 521-8818
Website: www.keystoneimed.com
Type of Test(s) Offered: OTC Rapid Antigen, PCR Lab
Cost: Free with Insurance or $125-$195
- Beeman’s RX Pharmacy
Address: 355 E. 21st St., San Bernardino 92404
Phone: (909) 882-3719
Website: www.beemans-rx.com
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with insurance or $9.99
- Beeman’s RX Pharmacy
Address: 399 E. Highland Ave. #103., San Bernardino 92404
Phone: (909) 886-6851
Website: www.beemans-rx.com
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with insurance or $9.99
- Waterman Pharmacy
Address: 2150 Waterman Ave. #104., San Bernardino 92404
Phone: (909) 882-5800
Website: www.watermanpharmacy.com
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $20
- Quick RX Pharmacy
Address: 590 N. Sierra Way., San Bernardino 92410
Phone: (909) 495-3599
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $20
- Delta Drugs Pharmacy
Address: 1666 Medical Center Dr., #1., San Bernardino 92411
Phone: (909) 887-7989
Website: www.deltadrugsrx.com
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home, Point of Care (POC), PCR take home/drop off
Cost: accepts most insurance $15-$35
- COVID Clinic San Bernardino (Drive-through)
Address: 500 Inland Center Dr., San Bernardino 92408
*Located in the parking lot outside Forever 21 near the intersection of N. Mall Way and S. E. St.
Phone: (877) 219-8378
Hours: Monday – Friday from 7 a.m. – 5 p.m. | Saturday from 9 a.m. – 5 p.m. | Sunday from 8 a.m. – 4 p.m.
Type of Test(s) Offered: Rapid Antigen, Rapid PCR, Point of Care (POC), Rapid Antibody
Cost: Free with Insurance or $75-$150
- Premier Urgent Care Center of California – San Bernardino
Address: 284 E. Highland Ave., San Bernardino 92404
Hours: 9 a.m – 7:30 p.m (Open 7 days a week)
Type of Test(s) Offered: POC Rapid Antigen, PCR Lab
Cost: Most Insurance accepted or $150-175
- King’s Pharmacy
Address: 1060 E. Foothill Blvd., #101, Upland 91786
Phone: (909) 982-1778
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: $16
- Ace Pharmacy
Address: 886 W. Foothill Blvd., #D, Upland 91786
Phone: (909) 946- 9400
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $12.99
- Mountain Plaza Pharmacy
Address: 1607 N. Mountain Ave., Upland 91786
Phone: (909) 985-0914
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $25
- ABC Pharmacy
Address: 954 W. Foothill Blvd., # D, Upland 91786
Phone: (909) 946-5512
Type of Test(s) Offered: Point of Care (POC), Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $10.99-$35.99
- Sav-on Pharmacy
Address: 1910 N. Campus Ave., Upland 91784
Phone: (909) 946-3340
Website: www.albertsons.com/pharmacy.html
Type of Test(s) Offered: Rapid Antigen, PCR Lab, Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $12
- Vons Pharmacy
Address: 81 W. Foothill Blvd., Upland 91786
Phone: (909) 981-1114
Website: www.vons.com/pharmacy.html
Type of Test(s) Offered: Rapid Antigen, PCR Lab, Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $12
- ADVANCED URGENT CARE CENTER
Address: 974 W. Foothill Blvd., Upland 91786
Phone: (909) 981-2273
Website: www.optumcare.com/ocnc/facilities/ca/upland/974-w-foothill-blvd
Type of Test(s) Offered: Rapid Antigen, PCR Lab
Cost: $70-$150
- Shima Hadidchi M.D. APC
Address: 1270 Hesperia Rd., Victorville 92395
Phone: (760) 999-0918
Website: shimahadidchimd.com
Type of Test(s) Offered: Rapid Antigen, PCR Lab
Cost: Free with insurance or $50-$80 or $120 if uninsured
- HealthRUs Urgent Care Walk-In
Address: 13622 Bear Valley Rd., Victorville 92392
Phone: (760) 261-4255
Website: healthrus.info
Type of Test(s) Offered: Rapid Antigen, PCR Lab
Cost: Free with insurance or $100-$150
- Clinica Mi Pueblo
Address: 14960 Bear Valley Rd., Victorville 92395
Phone: (760) 955-9300
Website: clinicamipueblo.com
Type of Test(s) Offered: Rapid Antigen, PCR Lab, Over-the-counter (OTC) Antigen take home
Cost: $50-$100
- Vons Pharmacy
Address: 12199 Hesperia Rd., Victorville 92395
Phone: (760) 241-8384
Website: www.vons.com/pharmacy.html
Type of Test(s) Offered: PCR Lab, Over-the-counter (OTC) Antigen take home
Cost: Free with insurance or $12
- COVID Clinic – The Mall of Victor Valley (Drive-through)
Address: 14400 Bear Valley Dr., Victorville 92397
* Located in the parking lot between Sears and JC Penney.
Hours: Monday – Saturday from 9 a.m. – 4:30 p.m., Sunday from 8 a.m. – 4 p.m.
Phone: (877) 219-8378
Type of Test(s) Offered: Rapid Antigen, Rapid PCR Lab, Point of Care (POC), Rapid Antibody
Cost: Free with Insurance or $75-$150
- Yucaipa Care Pharmacy
Address: 33490 Oak Glen Rd., Yucaipa 92399
Phone: (909) 570-9771
Type of Test(s) Offered: Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $12.99
- Vons Pharmacy
Address: 33644 Yucaipa Blvd., Yucaipa 92399
Phone: (909) 790-1961
Website: www.vons.com/pharmacy.html
Type of Test(s) Offered: PCR Lab, Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $12
- Vons Pharmacy
Address: 57590 29-Palms Hwy,. Yucca Valley 92284
Phone: (760) 365-0651
Website: www.vons.com/pharmacy.html
Type of Test(s) Offered: PCR Lab, Over-the-counter (OTC) Antigen take home
Cost: Free with Insurance or $12
Over-the-Counter (OTC) Antigen Test Kit Brands
Below is a list of some Over-the-Counter (OTC) Antigen test kits. You may find links to manufactures website, along with demonstration videos.
Please note, this is not a comprehensive list of all FDA authorized over-the-counter antigen test kits. Visit the FDA website for a list of authorized at home over-the-counter tests.
CareStart™
Result Time: 10 minutes
Demonstration video
FlowFlex™
Result Time: 15-30 minutes
Demonstration video
CLINITEST®
Result Time: 15 Minutes
Demonstration video
SCoV-2 Ag™
Result Time: 20 minutes
Demonstration video
iHealth®
Result Time: 15 minutes
Demonstration video
On/Go™
Result Time: 10 minutes
Demonstration video
QuickVue®
Result Time: 10 minutes
Demonstration video
FAQs
In San Bernardino County, we are dedicated to providing our community with continuity of COVID-19 services. We continue to provide over the counter COVID-19 test kits at no cost at any of our County facilitated test-kits distribution sites, this service will also continue as long as there is a supply. For those obtaining test kits through a medical provider, coverage will vary by insurance type.
- Medicare: People with traditional Medicare coverage will no longer receive free at-home tests.
- Medicaid: People with Medicaid coverage will have at-home tests covered at no cost through September 2024. After that date, COVID-19 at-home testing coverage will vary by state.
- Private insurance: People with private insurance and private Medicare plans (such as Medicare Advantage) will no longer be guaranteed free at-home tests. However, some insurers may continue to choose to cover them at their discretion.
- No insurance: People who are uninsured will continue to pay full price for at-home tests. Those who are uninsured or who cannot afford at-home tests may still be able to find them at a free clinic, community health center, public health department, library, or other local organization.
Similar restrictions will also apply to PCR and rapid antigen testing coverage. San Bernardino County sunset PCR testing in late December 2022.
- Medicare: People with traditional Medicare coverage will continue to receive free PCR and rapid antigen tests administered by a provider, but there may be a copay for the associated doctor’s visit.
- Medicaid: People with Medicaid will continue to receive free PCR and rapid antigen tests through September 2024. After that date, COVID-19 PCR and rapid antigen test coverage will vary by state.
- Private insurance: Coverage for PCR and rapid antigen tests will vary by insurer. People with private insurance and private Medicare plans may be subject to copays, depending on the plan. Some insurers may also begin to limit the number of covered tests or require tests to be done by in-network providers.
- No insurance: People who are uninsured can continue to purchase tests. Those who are uninsured or who cannot afford PCR and rapid antigen tests may still be able to access them at a free clinic or community health center.
The U.S. Food and Drug Administration (FDA) has authorized OTC antigen test under the Emergency Use Authorization (EUA).
To see a list of FDA approved OTC antigen tests, visit the FDA website here.
OTC antigen tests are typically 80% accurate in detecting an infected individual compared to PCR COVID-19 tests that have a 95% accuracy.
When using OTC antigen tests, the FDA recommends that a person repeats testing when receiving a negative test result; this is especially true for asymptomatic individuals. Findings have found that symptomatic individuals testing two times separated by 48 hours, increase the detection and accuracy of the SARS-COV-2 infection result by more than 90%. In asymptomatic individuals, repeated testing of 3 times every 48 hours, increases the test result accuracy by 79%.
To optimize results using a OTC antigen test, please ensure you follow the Instructions for Use found in your antigen test package and repeat testing as recommended.
For more information visit the FDA website.
OTC antigen test should never be frozen and typically require to be stored at room temperature (59-86 F). Please follow the manufactures instructions for use (IFUs) found in the package.
Below is a list of IFUs for some over-the-counter test kit brands:
Each test may be different, please refer to the manufacturer’s instruction for use typically found in the package.
Instruction videos are also available online:
CareStart™
FlowFlex™
iHealth®
On/Go™
CLINITEST®
American Sign Language-interpret positive self-test results
American Sign Language-interpret negative self-test results
**The County of San Bernardino does not sponsor or is affiliated with any organizations or manufacturers listed on this page.**
Watch How to Interpret Self-Test Results
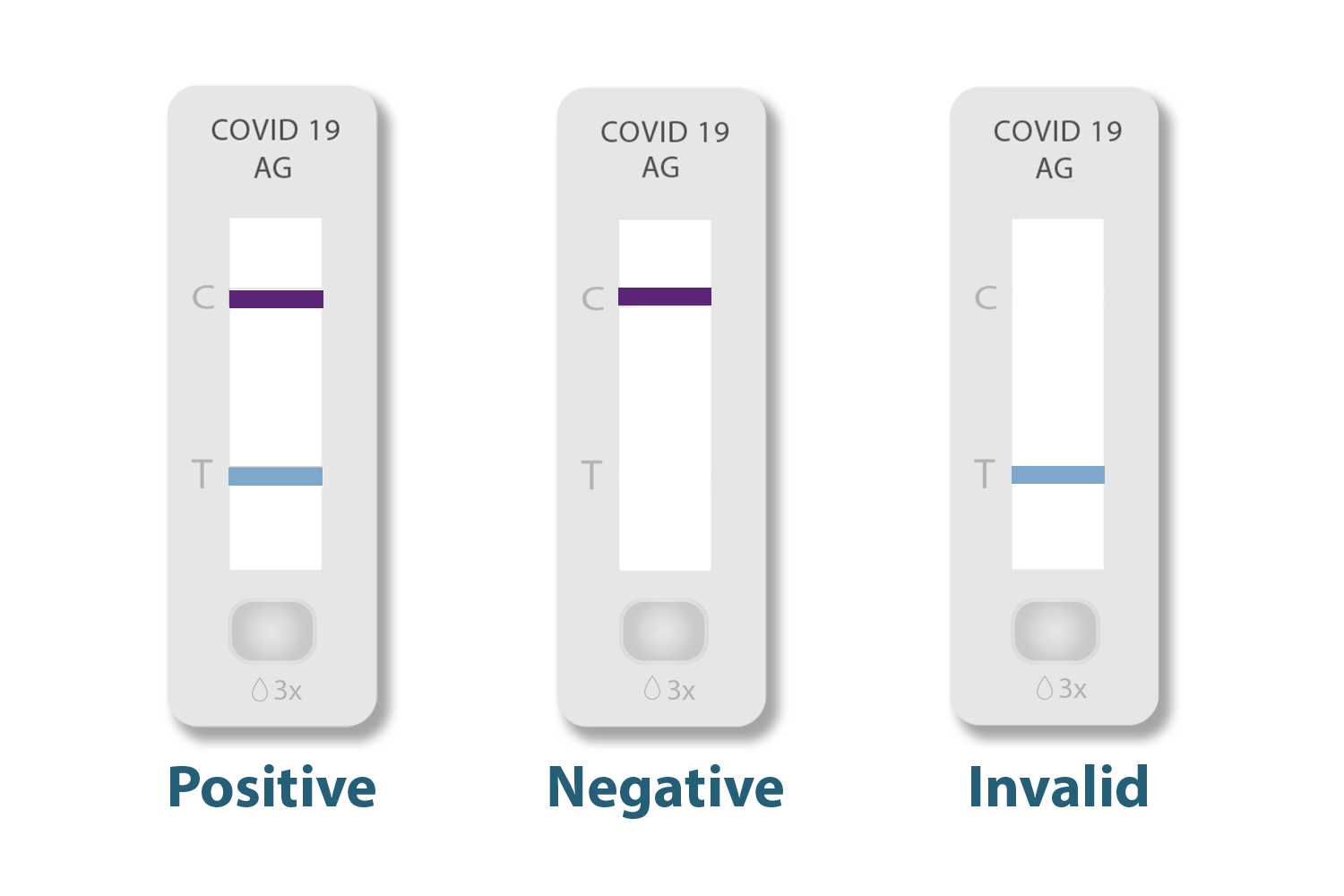
For the majority of each OTC antigen tests, results can be read as follows:
A positive result will have two lines, on both the “C” and “T.”
A negative result will only have one line next to the “C” and nothing next to the “T.’
An invalid result will either show a blank result window or a faded line next the “T” and nothing next to the “C.”
If you’re using the app, you take a picture of your test cassette to save your results – which can be used to show your doctor or others.
You have now completed your OTC antigen test! You can now safely dispose of all used test items into the trash.
A follow-up PCR COVID-19 test is recommended for those who test positive on a rapid antigen COVID-19 at-home test. To learn more about COVID-19 in San Bernardino County, please visit sbcovid19.com or call our COVID-19 Hotline at (909) 387-3911.
The primary role of case reporting is to allow public health agencies to take actions to mitigate disease spread.
Individuals should report their OTC antigen test result according to the instructions recommended by the test. If the test does not provide electronic reporting, positive test results should be shared with their healthcare provider in order to receive appropriate medical care.
Test results from a variety of OTC antigen tests can also be reported through learn.makemytestcount.org. MakeMyTestCount is a part of an effort at the National Institutes of Health to develop standard ways to report OTC antigen tests, and to securely and privately collect data in a way that makes it understandable. The results are then submitted to the same public health systems that currently receive COVID-19 results from tests done in labroratories and doctors’ offices.
While reporting OTC antigen test results is strongly encouraged, the US Government does not want to deter individuals from using self-tests. COVID-19 surveillance continues to be based on results from laboratory testing. The public health community, including CDC, is confident that situational awareness remains strong without receiving OTC test results.
Currently, the California Department of Public Health has authorized a blanket extension for the use of all FDA approved OTC antigen tests, even beyond their listed expiration dates or FDA extensions.
This emergency extension is temporary and may be discontinued by CDPH at any time. To stay up to date with the latest guidance, please refer to our Extensions Chart located on the top right side of the page.
(Information revised on 11/14/22)
Yes, fully vaccinated people who have come into close contact with someone with COVID-19 should be tested on Day 5-6 following exposure, even if no symptoms develop.
Because you can still develop COVID-19 after 10 days of being exposed, wear a mask to take precautions and protect others.
Please refer to the CDC, for more guidelines following exposure or to see the latest quarantine and isolation information.
NOTE: These locations offer OTC antigen tests and should only be considered options. It is not a comprehensive list of all locations offering COVID-19 antigen testing. Amazon and Walmart are currently offering OTC antigen tests at cost.
CVS PHARMACY
Contact Information: https://www.cvs.com/minuteclinic/covid-19-testing
Cost: Free
Location Information: Per CVS – COVID-19 testing is performed at no cost to you. Health insurance companies are required to cover 100% of the cost. If you have insurance, we will ask for those details. If not, we may ask for your Social Security number or driver’s license/state ID in order to bill the federal program for the uninsured. In either case, you will owe nothing out of pocket.
PREMIER URGENT CARE CENTER OF CALIFORNIA – SAN BERNARDINO
Contact Information: https://www.premierurgentcarecalifornia.com/covid-19-testing or call (909) 883-1098
Cost: $150.00
Location Information: Walk-in only. Antigen testing only. Locations in Indio, Coachella, San Bernardino and Riverside. Insurance may cover the testing cost. They offer PCR testing as well.
TOTALCARE (WALK-IN CLINIC) – Chino
Contact Information: https://totalcarenow.com/covid/
Cost: $80.00
Location Information: Locations in Chino, Eastvale, Corona, El Monte. Rapid test is self-pay only.
ONE TESTING
Contact Information: Call (909) 360-8818
Cost: $80.00
Location Information: Locations in Chino, Eastvale, Corona, El Monte. Rapid test is self-pay only.
CENTRAL URGENT MEDICAL CARE
Contact Information: Call (909) 941-0920
Cost: $125.00
Location Information: 9695 Baseline Rd., Rancho Cucamonga
ABOVE URGENT CARE
Contact Information: Call (909) 297-3361
Cost: $120.00
Location Information: 8891 N. Central Ave., Suite A, Montclair
HEALTH R US
Contact Information: Call (760) 261-4255
Cost: $100.00
Location Information: 13622 Bear Valley Rd., Suite 9, Victorville
Below are a few online and retail purchase options for OTC antigen tests.
Abbott BINAXNow
Result Time: 15 minutes
Test Demonstration/Directions: HOW TO: A Guide for the BinaxNOW™ COVID-19 Self Test
Access Bio CareStart
Result Time: 10 minutes
Test Demonstration/Directions: Method of Use
Ellume COVID-19 Home Test
Result Time: 15 minutes
Test Demonstration/Directions: How It Works
Orasure Intelliswab Test
Result Time: 30 minutes
Test Demonstration/Directions: Instructions for Use
Quidel QuickVue At-Home
Result Time: 10 minutes
Test Demonstration/Directions: User Instructions
Medek aiDX Rapid
Result Time: 10 minutes
Test Demonstration/Directions: Method of Use
To learn more about OTC antigen testing, please visit the CDC interim guidance for more information.
